|
This is because electrons distribute around an atom to minimize repulsion with one another. By knowing the electron domain of each atom in a molecule, you can predict its geometry. It represents the number of locations expected to contain electrons. The molecule BrF3 has an) electron-domain geometry. The molecule AsF3 has a (n) electron-domain geometry. The hybridization of phosphorous in PCl 5 is sp 3 d. The electron geometry of PCl 5 is also Trigonal bipyramidal. The molecule CH3 has a (n) electron-domain geometry. In the PCl 5 Lewis dot structure, a total of 15 lone pairs and 5 bond pairs are present. Question: The molecule XeF2 has a (n) electron-domain geometry. Now that we know the valence electrons in the molecule, we can start with making the Lewis diagram for the compound. Hello GuysBromine Trifluoride or BrF3 is an interhalogen compound consisting of two halogens - Bromine and Fluorine atoms. An atom's electron domain is the number of lone pairs or chemical bond locations that surround it. This problem has been solved You'll get a detailed solution from a subject matter expert that helps you learn core concepts. The valence shell electron pair repulsion (VSEPR) model focuses on the bonding and nonbonding electron pairs present in the outermost (valence) shell of an atom that connects with two or more other atoms.įundamentally, the VSEPR model theorizes that these regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. Br 7 electrons F 73 14 electrons ( as there are three fluorine atoms, we will multiply the number of valence electronstoo) Hence the total number of valence electrons for BrF3 is 28. VSEPR Theory: a chemistry model used to predict the shape of individual molecules based on electron-pair electrostatic repulsion.Start your trial now First week only 4.99 arrow. The main geometries without lone pair electrons are: linear, trigonal, tetrahedral, trigonal bipyramidal, and octahedral. Solution for Predict the electron-domain and molecular geometries of(a) BrF3, (b) SF5+.BrF3 electron geometry BrF3 Hybridization BrF3 Bond angle Is BrF3 polar BrF3 electron. Place the following in order of increasing X-A-X bond angle, where A represents the central atom and X represents the outer atoms in each molecule. Molecular Geometry of BrF3, Trigonal Bipyramidal. Trending now This is a popular solution Step by step Solved in 2 steps with 1 images. 
Give the molecular geometry and number of electron groups for BrF3. ( Select According to VSEPR theory, what is the electron domain geometry of BRF3 V Select tetrahedral see-saw trigonal bipyramidal trigonal planar bent linear us Next square pyramidal Expert Solution. This is due to the two lone pairs of electrons on bromine, which cause a repulsion from the fluorine. A molecule with a square pyramidal molecular geometry has a bond angle of. 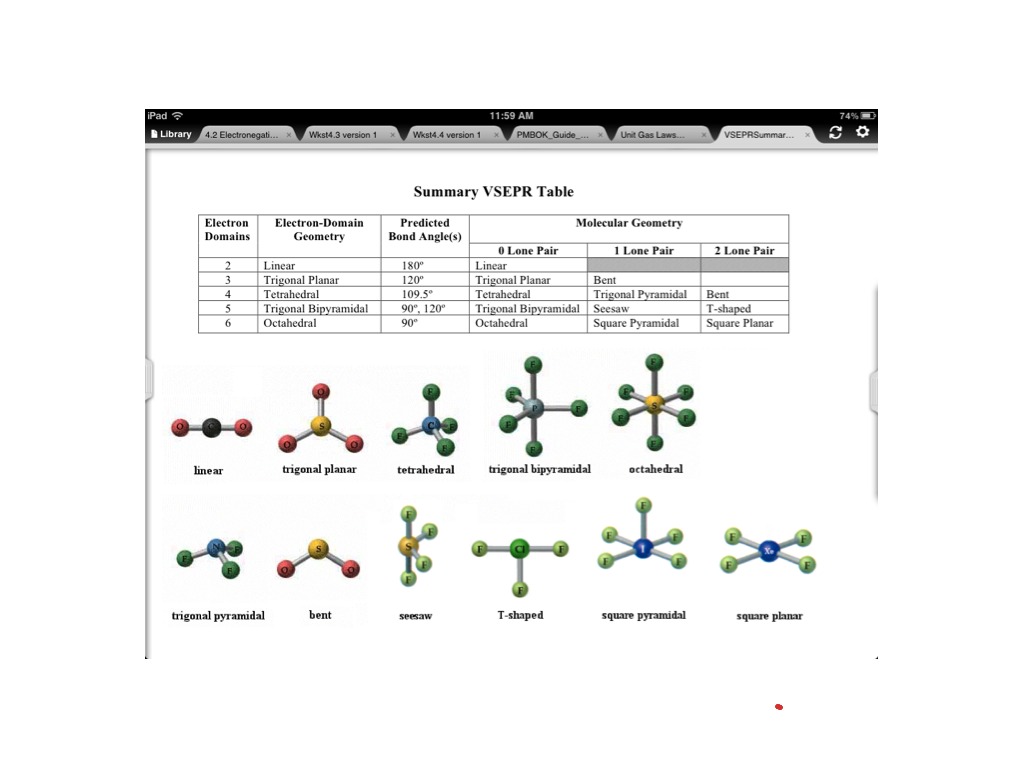
Molecular geometries take into account the number of atoms and the number of lone pair electrons. The preferred geometry of molecule BrF3 or bromine trifluoride is a planar 'T-shaped' molecule.Fundamentally, the VSEPR model theorizes that regions of negative electric charge will repel each other, causing them (and the chemical bonds that they form) to stay as far apart as possible. 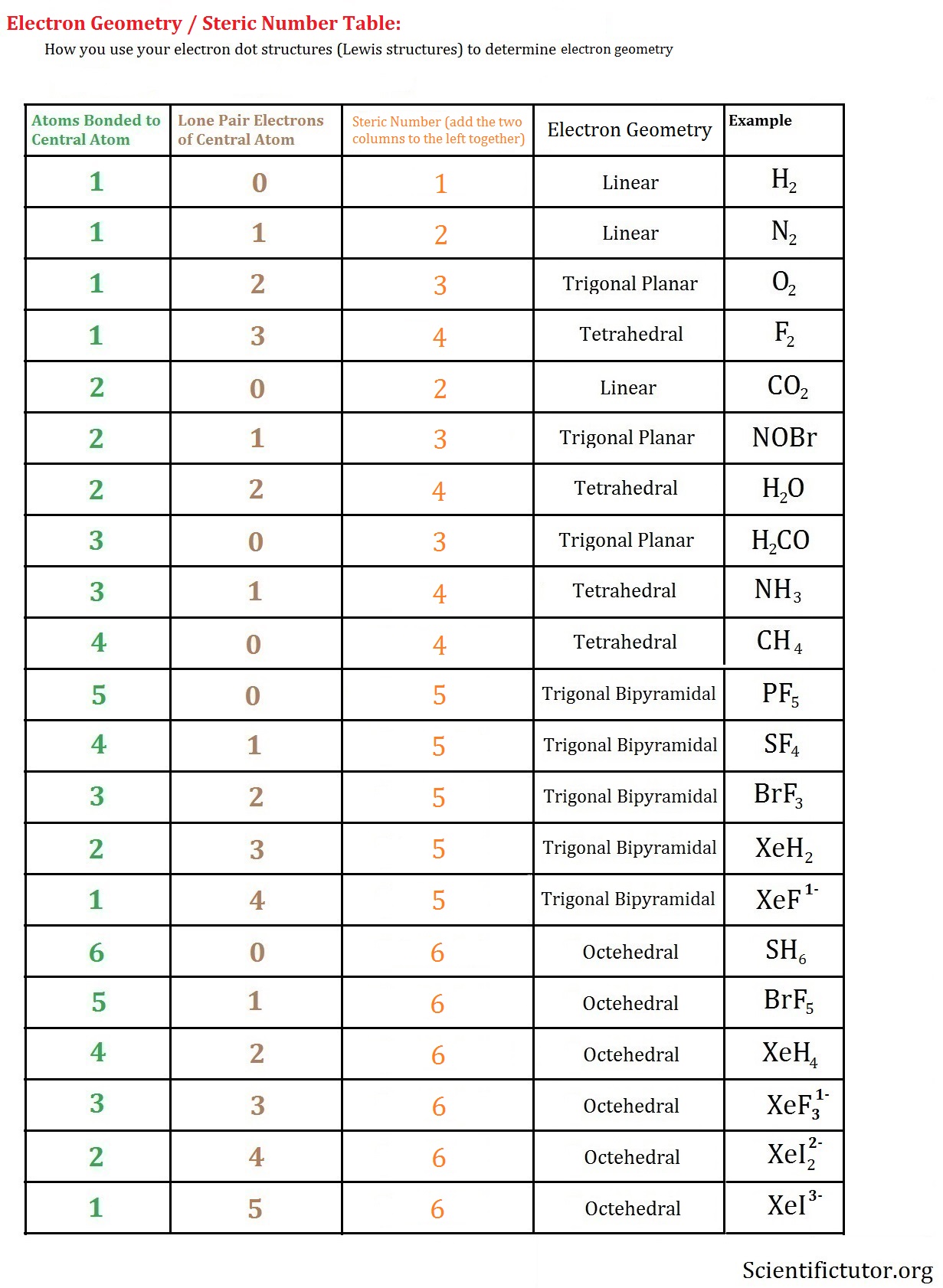 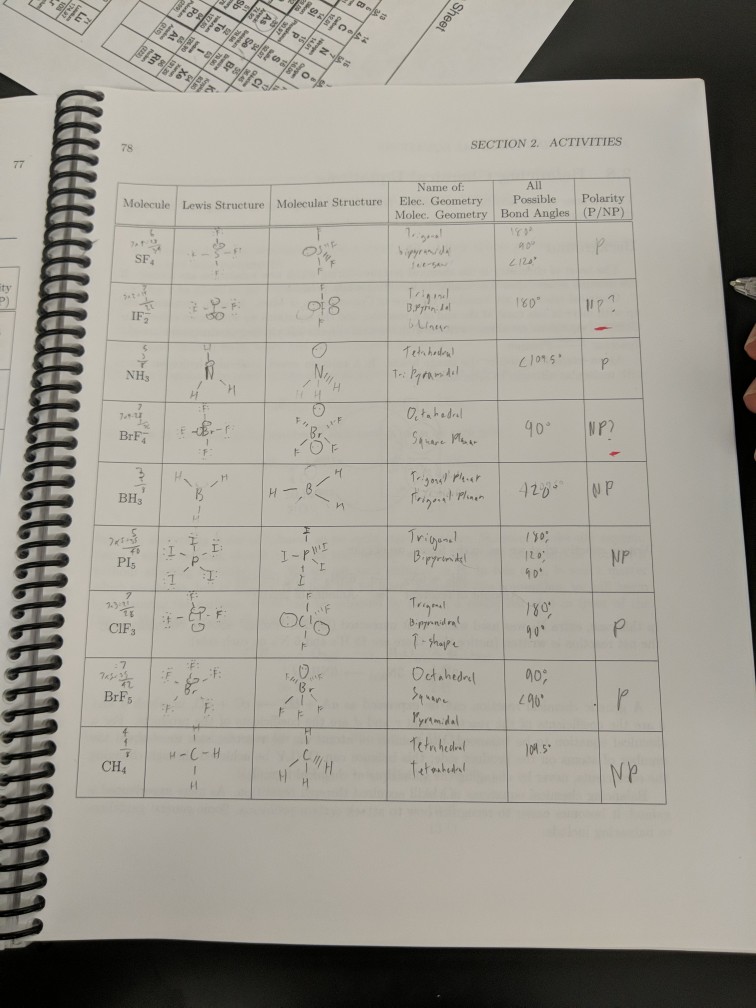
Trending Questions How many pounds is 14.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
